Bria-IMT™ Cell Therapy
Latest Developments
BriaCell Pivotal Phase 3 study of Bria-IMT™ in metastatic breast cancer is ongoing
The pivotal Phase 3 combination study is listed on ClinicalTrials.gov as NCT06072612.
Bria-IMT™ was awarded Fast Track status by the U.S. Food and Drug Administration (FDA).
About Bria-IMT™ (SV-BR-1-GM)
Developed and characterized by a team of dedicated scientists and clinicians, Bria-IMT™ (SV-BR-1-GM) is an off-the-shelf targeted cell-based immunotherapy for the treatment of metastatic breast cancer (i.e. breast cancer that has spread from the breast or armpit to other areas of the body including the bones, lungs, liver or sometimes the brain). Bria-IMT™ is a genetically engineered human breast cancer cell line with features of immune cells. It is clinically used as a targeted immunotherapy.
Mechanism of Action of Bria-IMT™: The mechanism of action of Bria-IMT™ is currently under investigation. We hypothesize that Bria-IMT™ both indirectly and directly stimulates the body’s own cancer-fighting cells to attack and destroy breast cancer tumors. INDIRECTLY, Bria-IMT™ produces multiple breast cancer “antigens” (proteins that are expressed in breast cancer cells) that are identified by the patient’s immune system. Some of these same antigens are expressed by a patient’s breast cancer and are recognized by the immune system which activates antigen specific tumor killing cells. In addition, Bria-IMT™ appears to be able to DIRECTLY stimulate these cancer-fighting immune cells markedly, further boosting the body’s immune cell response.
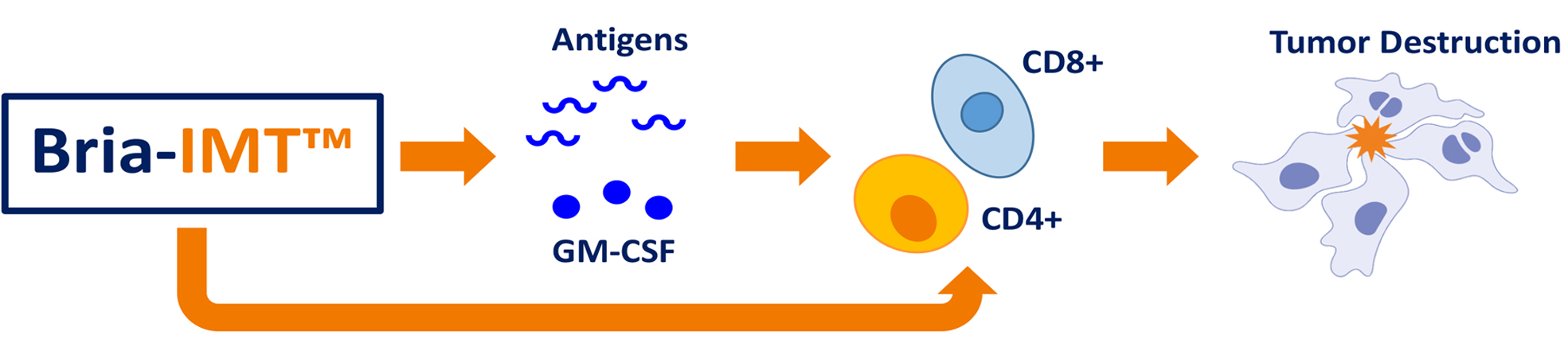
We believe that Bria-IMT™:
2. The immune cells present the antigens to CD4+ and CD8+ T-cells, cells known for tumor destruction.
3. Secretes a protein called GM-CSF which further boosts the immune response.
4. Also directly stimulates cancer-fighting T-cells, acting like an immune cell, further boosting the immune cell’s response.
Why Bria-IMT™ Is Different
Though breast cancer treatment has come a long way in recent years, many patients — especially those with advanced or treatment-resistant disease — still face limited options. BriaCell’s Bria-IMT™ breast cancer immunotherapy offers a groundbreaking approach for patients with breast cancer that addresses some of the biggest challenges in current care. Here’s what sets BriaCell apart from traditional cancer treatments.
Off-the-Shelf Convenience
Most cell-based therapies require doctors to harvest and return a patient’s cells — something that takes valuable time. However, the Bria-IMT skips this step to deliver off-the-shelf immunotherapy for breast cancer that’s readily available without delays or the complexity and expense of cell harvesting. This speeds up the start of BriaCell cancer therapy when time matters most.
Works Well With Other Immunotherapies
Bria-IMT doesn’t work in isolation. This targeted call-based therapy teams up with other cutting-edge treatments like checkpoint inhibitors. These therapeutic combinations work to remove barriers that cancer puts up to hide from the immune system. Once Bria-IMT and its companion therapies break through, they amplify the body’s ability to detect and destroy cancer cells.
Promising Clinical Trial Results
For patients who have already tried and exhausted multiple treatments, early trial results for Bria-IMT offer hope. Recent studies showed a longer overall median survival rate in a specific group of advanced breast cancer patients when compared to traditional treatments. The therapy has also been very well tolerated, with no patients discontinuing therapy due to side effects from Bria-IMT treatment. This data shows promise for patients with breast cancer most in need of new solutions.
Who Might Benefit From Bria-IMT?
Designed with specific patient groups in mind, Bria-IMT provides immunotherapy for those who haven’t found success with other options. Patients with advanced or metastatic breast cancer, especially those whose disease has spread beyond the breast, require more intensive treatment options. Many face limited therapeutic choices for advanced breast cancer treatment, but Bria-IMT may enable care to move in a new direction.
Many patients receiving Bria-IMT treatment in clinical trials have already tried several therapies without success. For those patients, our novel cancer immunotherapy offers a unique approach and renewed potential. Our targeted cell-based therapy often proves crucial when conventional methods no longer work for patients with breast cancer.
Bria-IMT and BriaCell’s Mission
From our clinical strategies to our commitment to making treatments more widely available, BriaCell delivers a transformable approach to breast cancer treatment, one patient-first innovation at a time. The patient always comes first at BriaCell, and this philosophy fuels our commitment to rethinking breast cancer care and developing effective, accessible therapies. Bria-IMT embodies this vision, paving the way for safer, smarter immunotherapy solutions.
Rooted in transforming how we treat breast cancer, we offer cell-based immunotherapies that provide patients with safer, more convenient treatments. BriaCell cancer therapy goes beyond helping patients with breast cancer — we’ve created a growing pipeline for treatments that includes Bria-OTS™. This customizable off-the-shelf immunotherapy platform further broadens patient access to cancer care.

